Investors welcome a study for a possible vaccination against the corona virus by Pfizer and BioNTech. As a result, Pfizer’s shares have risen sharply. Hopes for a vaccine against the SARS-COV2 virus are expected and supported in October. Even if the vaccine is available, the logistics of producing and effectively distributing such a vaccine on a global scale or even with large countries is rarely discussed.
Vaccine manufacturers already produce hundreds of millions of doses
Currently, multiple vaccine candidates are in so-called at-risk production. Vaccine producers have started producing hundreds of millions of doses to be ready for distribution once a candidate passes phase III trials. While the produces bear some of the financial burden should the candidate fail, most of the cost will fall onto governments (U.S. through BARDA in case of the Oxford vaccine) and NGOs (Gates Foundation). There is also the question of crowding out other vaccine production chains, an issue affecting primarily emerging economies.
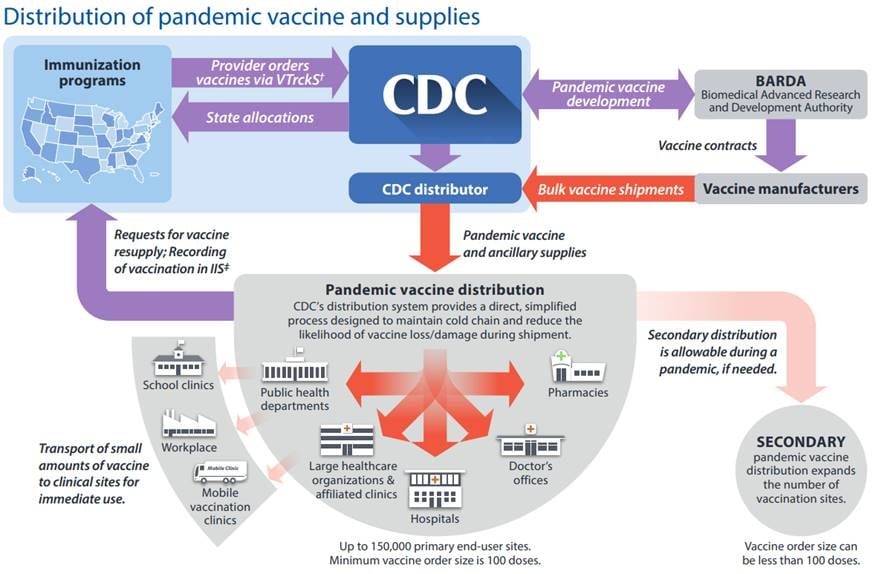
https://espanol.cdc.gov/flu/pdf/pandemic-resources/pandemic-influenza-vaccine-distribution-9p-508.pdf Note: Past performance is not indicative of future development.
Worldwide distribution of a vaccine a huge task
Distributing a vaccine globally and even in large countries is a monumental task and requires rigorous governmental planning and monitoring. Most developed economies have strategic playbooks for pandemics, however, these were often written with a flu pandemic in mind. They regulate who gets the vaccine first with the general aim being the prevention of loss of life and public order. Therefore, vulnerable groups, medical personnel, police, armed forces and central government will be vaccinated first. The distribution to the broad population is then governed by the logistics in place for such an operation. For example, in the U.S. the Center for Disease Control and Prevention (CDC) is the only institution that can provide cold chain logistics and monitoring of a nationwide vaccine distribution.
While the CDC has been heavily criticized during the current pandemic for its botched handling of tests, concerns for a similar mess-up during vaccine distribution should be weighed against the successful distribution of the swine flu vaccine in 2009. Another concern, however, is public acceptance of vaccine. While so called anti-vaxxers pose a small, albeit loud, proportion of the population, many people have voiced concerns of getting a vaccine they perceived was rushed through development. Most government guidelines state the urgent need for public leaders to address these concerns with consistent and transparent education. To illustrate the issue that at least the U.S. might face see below:
Supply chain monitoring essential
An often underestimated task during a large vaccination campaign is monitoring, which ensures that the right amount of doses arrive at the right destination, vaccines are not sitting idle in distribution centers or end up in the black market, adverse side effects are caught early and most importantly the government knows who has been vaccinated. A national vaccination campaign requires an effective bureaucracy apparatus and transparent communication between the involved government agencies. Cyberdefence is an often neglected factor in this phase, as bad actors could potentially seriously interfere with the vaccination campaign at this point with relatively simple means.
Conclusion
The hope for a possible vaccine in October supports the positive development on the stock markets. In addition to monetary and fiscal policy measures, a remedy against Covid19 would be the right medicine for a continuation of the upward trend.
Interesting Links
Online calculator for health care system utilization
Nice tool to simulate the epidemic course of COVID19
Tool for tracking gene mutations in COVID19 and their local occurrence
Risk-map for the U.S. with various risk overlays
New York Times: Tracking the true toll of the Coronavirus Crisis
New York Times: Vaccine Tracker
Steckbrief COVID-19 Robert Koch Institut (German):
The Robert Koch Institute (RKI) is a German federal institute of the Ministry of Health for disease surveillance and prevention. The summarize the findings of available research about e.g.
Mortality rate, how long is someone infectious, time of hospitalization,…
Government Response Tracker:
The Oxford COVID-19 Government Response Tracker counts data from 73 countries. It is designed to systematically record government responses worldwide and aggregate the scores into a common ‘Stringency Index’.
https://www.bsg.ox.ac.uk/research/research-projects/oxford-covid-19-government-response-tracker
Podcast about the Corona virus (German):
Prof. Dr. Christian Drosten, Director of the Institute for Virology at the Charité
Topics: Corona virus in everyday life, dangers for children, health care,…
How many Covid-19 Tests are performed?
USA Daily new confirmed cases:
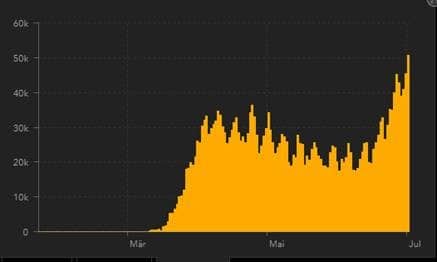
Source: https://gisanddata.maps.arcgis.com/apps/opsdashboard/index.html#/bda7594740fd40299423467b48e9ecf6
Legal note:
Prognoses are no reliable indicator for future performance.
Legal disclaimer
This document is an advertisement. Unless indicated otherwise, source: Erste Asset Management GmbH. The language of communication of the sales offices is German and the languages of communication of the Management Company also include English.
The prospectus for UCITS funds (including any amendments) is prepared and published in accordance with the provisions of the InvFG 2011 as amended. Information for Investors pursuant to § 21 AIFMG is prepared for the alternative investment funds (AIF) administered by Erste Asset Management GmbH pursuant to the provisions of the AIFMG in conjunction with the InvFG 2011.
The currently valid versions of the prospectus, the Information for Investors pursuant to § 21 AIFMG, and the key information document can be found on the website www.erste-am.com under “Mandatory publications” and can be obtained free of charge by interested investors at the offices of the Management Company and at the offices of the depositary bank. The exact date of the most recent publication of the prospectus, the languages in which the fund prospectus or the Information for Investors pursuant to Art 21 AIFMG and the key information document are available, and any other locations where the documents can be obtained are indicated on the website www.erste-am.com. A summary of the investor rights is available in German and English on the website www.erste-am.com/investor-rights and can also be obtained from the Management Company.
The Management Company can decide to suspend the provisions it has taken for the sale of unit certificates in other countries in accordance with the regulatory requirements.
Note: You are about to purchase a product that may be difficult to understand. We recommend that you read the indicated fund documents before making an investment decision. In addition to the locations listed above, you can obtain these documents free of charge at the offices of the referring Sparkassen bank and the offices of Erste Bank der oesterreichischen Sparkassen AG. You can also access these documents electronically at www.erste-am.com.
Our analyses and conclusions are general in nature and do not take into account the individual characteristics of our investors in terms of earnings, taxation, experience and knowledge, investment objective, financial position, capacity for loss, and risk tolerance. Past performance is not a reliable indicator of the future performance of a fund.
Please note: Investments in securities entail risks in addition to the opportunities presented here. The value of units and their earnings can rise and fall. Changes in exchange rates can also have a positive or negative effect on the value of an investment. For this reason, you may receive less than your originally invested amount when you redeem your units. Persons who are interested in purchasing units in investment funds are advised to read the current fund prospectus(es) and the Information for Investors pursuant to § 21 AIFMG, especially the risk notices they contain, before making an investment decision. If the fund currency is different than the investor’s home currency, changes in the relevant exchange rate can positively or negatively influence the value of the investment and the amount of the costs associated with the fund in the home currency.
We are not permitted to directly or indirectly offer, sell, transfer, or deliver this financial product to natural or legal persons whose place of residence or domicile is located in a country where this is legally prohibited. In this case, we may not provide any product information, either.
Please consult the corresponding information in the fund prospectus and the Information for Investors pursuant to § 21 AIFMG for restrictions on the sale of the fund to American or Russian citizens.
It is expressly noted that this communication does not provide any investment recommendations, but only expresses our current market assessment. Thus, this communication is not a substitute for investment advice.
This document does not represent a sales activity of the Management Company and therefore may not be construed as an offer for the purchase or sale of financial or investment instruments.
Erste Asset Management GmbH is affiliated with the Erste Bank and austrian Sparkassen banks.
Please also read the “Information about us and our securities services” published by your bank.


